
Nuclear fission subdivision of a heavy atomic nucleus such as that of uranium or plutonium into two fragments of roughly equal mass. For fusion the energy release occurs when the mass of the fusion products is smaller than the reactants. How do the products of a nuclear fusion reaction compare with the reactants?įusion of light elements (the reactants) into heavier elements (the products) releases energy (as does fission of heavy elements into lighter elements). The fusion fuels deuterium and helium (the heavy forms of hydrogen) fuse into helium releasing a high energy neutron. Which of the following are the products of the fusion of hydrogen and deuterium? The larger nuclei again needs less energy to hold it together – so energy is released. … Fusion on the other hand is the process of sticking together light nuclei (typically hydrogen -like nuclei). How does fusion and fission produce energy?įission is the splitting of heavy nuclei (such as uranium) – in two smaller nuclei. … The heat is used to make steam to produce electricity. When the nucleus of a U-235 atom captures a moving neutron it splits in two (fissions) and releases some energy in the form of heat also two or three additional neutrons are thrown off. How are the products of the fission of U 235 transformed into household energy such as electricity? The uranium-235 absorbs the neutron and forms an unstable compound nucleus uranium-236. The absorption of a “slow” neutron induces the fission of a uranium-235 nucleus. How is the fission of a uranium-235 nucleus induced? During fission the nucleus of the atom splits apart producing both heat and extra neutrons. U 235 when concentrated (or “enriched”) is fissionable in light-water reactors (the most common reactor design in the USA).
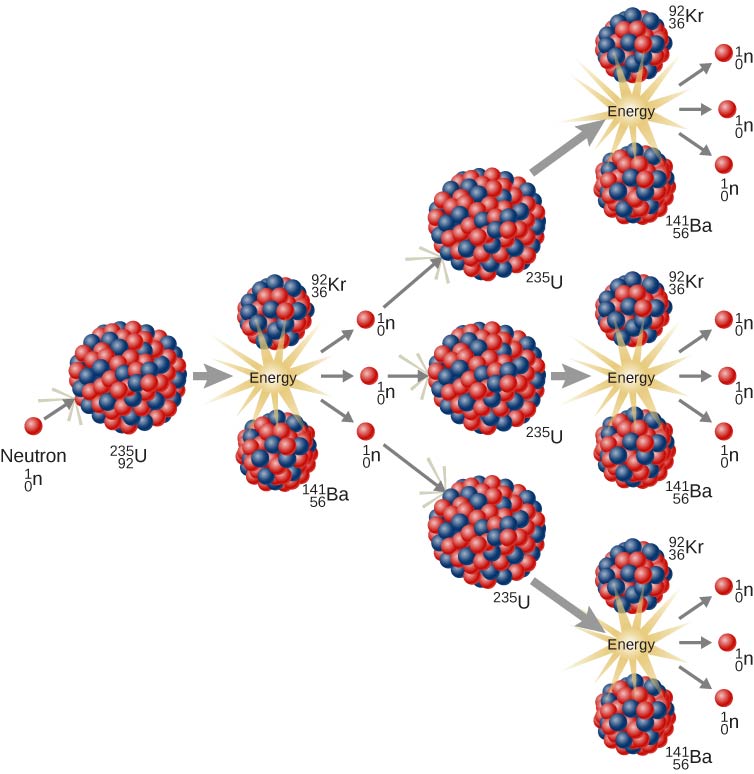
Typically when the plutonium 239 nucleus undergoes fission the nucleus splits into two smaller nuclei (triple fission can also rarely occur) and a few neutrons (the average is 2.89 neutrons per fission by thermal neutron) and release of energy in the form of heat and gamma rays. Some of these neutrons are absorbed by other atoms of uranium-235. When a nucleus of uranium-235 undergoes fission it splits into two smaller atoms and at the same time releases neutrons ( n) and energy. Nuclear power plants currently in use depend primarily on the fission of uranium-235 and plutonium-239. The reaction also releases three neutrons and a great deal of energy. As a result it splits into two smaller nuclei krypton-92 and barium-141. In either case the nucleus of uranium-235 becomes extremely unstable with the extra neutron. What two elements are created after the fission of uranium? The two smaller nuclei are the fission products. Typically a large nucleus like that of uranium fissions by splitting into two smaller nuclei along with a few neutrons the release of heat energy (kinetic energy of the nuclei) and gamma rays. What is formed from the fission of uranium? It was discovered in 1935 by Arthur Jeffrey Dempster. … Uranium-235 has a half-life of 703.8 million years. Unlike the predominant isotope uranium-238 it is fissile i.e. Uranium-235 ( 235U) is an isotope of uranium making up about 0.72% of natural uranium.
